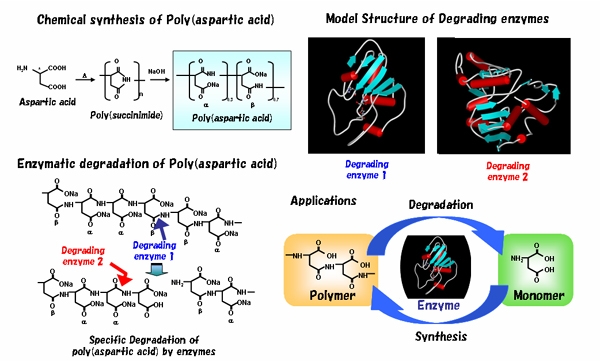
Enzymatic synthesis of novel polymer materials
|
Polymer thin film is
prepared as a functional membrane on the substrate surface of a biosensor to
measure the interaction between the membrane and molecules. Poly(aspartic acid),
which has functional carboxyl groups, shows high biocompatibility and
biodegradability. We are studying the enzymatic synthesis of poly(aspartic
acid) with well-controlled structure.
|
References
|
(1) |
T. Hiraishi, Y. Hirahara, Y. Doi, M.
Maeda, and |
|
(2) |
Y. M. Normi, T. Hiraishi, S. Taguchi, H.
Abe, K. Sudesh, N. Najimudin, Y. Doi, “Characterization and Properties of G4X Mutants of Ralstonia
eutropha PHA synthase for Poly(3-hydroxybutyrate) Biosynthesis in Escherichia
coli.”, Macromol. Biosci., 5, 197-206 (2005) |
|
(3) |
Y. M. Normi, T. Hiraishi, S. Taguchi, K.
Sudesh, N. Najimudin, Y. Doi, “Site-directed Saturation Mutagenesis at Residue F420 and Recombination
with Another Beneficial Mutation of Ralstonia eutropha PHA synthase.”, Biotechnol. Lett., 27, 705-712 (2005) |
|
(4) |
Y. Kikkawa, K. Yamashita, T. Hiraishi, M.
Kanesato, Y. Doi, “Adsorption
Behavior of Poly(3-hydroxybutyrate) Depolymerase on Polyester Surface
Investigated by QCM and AFM.”, Biomacromolecules, 6,
2084-2090 (2005) |
|
(5) |
T. Hiraishi, Y. Kikkawa, M. Fujita, N. M.
Yahaya, M. Kanesato, T. Tsuge, K. Sudesh, M. Maeda, Y. Doi, “AFM Observation of In Vitro
Polymerized Poly[(R)-3-hydroxybutyrate] : Insight into Possible Mechanism of
PHB Granule Formation.”, Biomacromolecules, 6,
2671-2677 (2005) |
|
(6) |
Y. Kikkawa, M. Narike, T. Hiraishi, M.
Kanesato, K. Sudesh, Y. Doi, T. Tsuge, “Organization of Polyhydroxyalkanoate Synthase for In Vitro
Polymerization as Revealed by Atomic Force Microscopy.”, Macromol. Biosci., 5, 929-935 (2005) |
|
(7) |
T. Hiraishi, and Y. Doi, “Enzymatic degradation of
poly(aspartic acid) catalyzed by its hydrolases.”,
Bio Industry, 22, 43-50 (2005) |
|
(8) |
T. Hiraishi, M. Kajiyama, |
|
(9) |
T. Hiraishi, M. Kajiyama, K. Tabata, H.
Abe, |
|
(10) |
T. Hiraishi, M. Kajiyama, K. Tabata, |
|
(11) |
T. Hiraishi, K. Tabata, and Y. Doi, “Microbial and Enzymatic Hydrolysis
of Poly(aspartic acid).” RIKEN Review, 42, 81-84
(2001) |
|
(12) |
K. Tabata, M. Kajiyama, T. Hiraishi, H.
Abe, |